The LOGIC project scales-up the Liquid-Out Gas-In Concept (LOGIC) reactor for methanol synthesis from CO2 and sustainable hydrogen.
In short:
- Storage of sustainable energy for longer periods becomes increasingly important
- There is a rising demand for sustainable carbon feedstock
- LOGIC aims to combine these 2 by producing methanol from CO2 and H2 at mild temperatures and pressures with a carbon yield of nearly 100%
Storing sustainable energy
Because of the rising amount of intermittently available sustainable energy, such as sustainable hydrogen, the storage of this energy for longer periods becomes increasingly important. Furthermore, there is a rising demand for sustainable carbon feedstocks in the chemical industry.
The LOGIC technology aims to combine these two factors by providing a process for producing methanol from CO2 and H2 at mild temperatures and pressures with a carbon yield of nearly 100%. This is realized by using an innovative reactor design with an internal gas recycle.
In addition, an in-situ condenser removes the condensible methanol and water from the reactor while allowing the gases to be recycled. Another advantage of the process is the possibility of operating without external heat input and with natural convection driving the internal gas circulation.
Proof-of-principle reactor
In previous work at the University of Twente a proof-of-principle reactor was successfully built and tested. Experiments showed a methanol yield of nearly 100%. Furthermore it was shown that the reactor is able to operate under natural convection conditions. Modelling efforts showed the possibility of autothermal operation if sufficient heat integration is applied.
Objectives of the LOGIC project
- To scale-up the LOGIC reactor from the previously built proof-of-principle reactor to a 5 kg/day pilot unit. Build, test and optimize the scaled-up reactor
- Investigate the natural convection phenomena in the system in combination with autothermal operation of the reactor.
- Make a design for a large-scale unit.
Approach
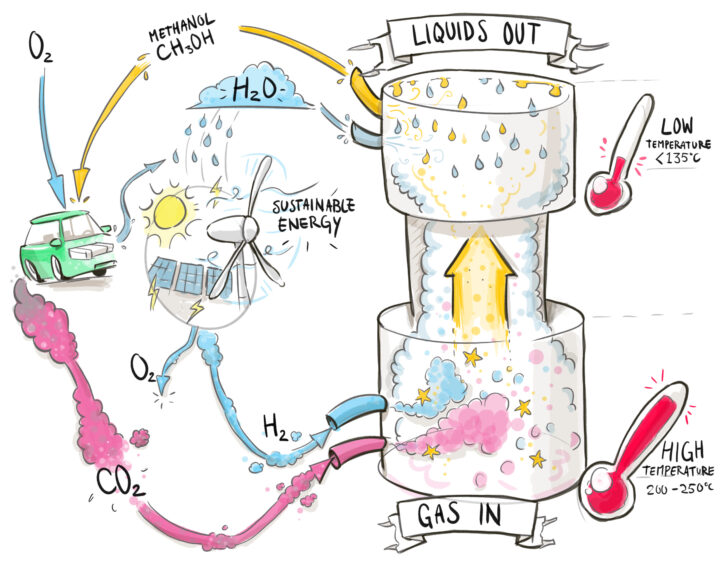
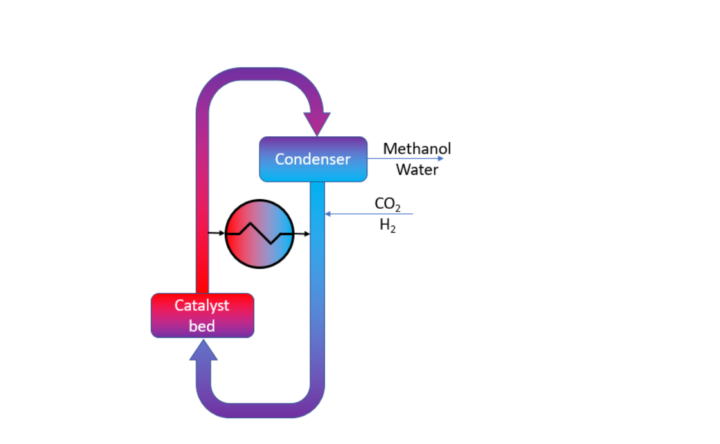
The LOGIC concept works as follows (see also Figure 1):
- CO2 and H2 are fed into the reactor in a molar ratio of 1:3 and mixed with the recycle gas stream.
- A heat exchanger heats up the fresh gas using heat from the catalyst outlet stream.
- The exothermic reaction takes place inside the catalyst bed, heating up the gas.
- Heat from the catalyst outlet is exchanged with the catalyst feed stream.
- Water and methanol are condensed by locally lowering the temperature and are removed from the reactor.
- The unconverted gases are recycled.
Both experimental work as well as modelling was performed into the fundamental aspects of the system. Intrinsic reaction kinetics were determined and the interplay between gas circulation and heat exchange under natural convection conditions was studied. A prototype (design capacity 10 kg/day) was designed and built early in the project and tested under various conditions. The results were compared with those of the developed models. Based on experimental work, new insights are obtained and models are developed. With these it is found that the reactor can operate autothermal, when heat losses to the environment are strongly reduced. Furthermore, the dynamic behaviour at start-up, shut down and process variations is studied, also in view of intermittent operation. Based on the results of the (validated) models and the prototype, a design for a 100 kg/h installation was made.
Obtained results and outlook
The project delivered a validated, scalable, new technology for the (large and small-scale) standalone production of methanol from (renewable) CO2 and H2. The concept was validated on a scale of approx. 10 kg/day with a working prototype. Although developed with a view to stand-alone methanol production from sustainably obtained CO2, H2 and renewable electricity, the concept is also applicable on a larger scale. Moreover, in addition to renewable CO2 from ambient air or biogas, the technology can also be used for mixtures of CO2 and CO and in principle also for other, equilibrium-limited reactions of gaseous components to condensable liquid products (such as ammonia).
You might also be interested in
Acknowledgement & partners
This project is co-funded with subsidy from the Topsector Energy by the Ministry of Economic Affairs and Climate Policy.