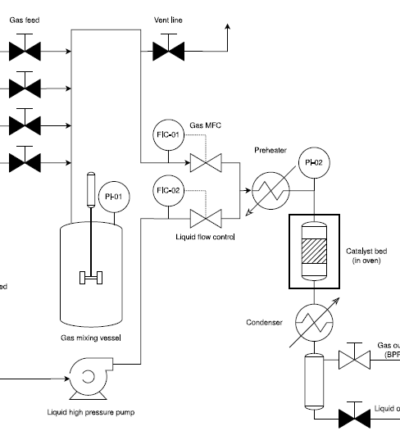
The present work introduces a new kinetic model for methanol synthesis using a Cu/ZnO/Al 2O 3 catalyst, a process important for reducing CO2 emissions and producing useful chemicals. By incorporating the latest insights from literature on the mechanisms, an extensive many-parameter model was derived from elementary adsorption and reaction steps. The model was checked against experimental data from literature and an extensive state-of-the-art set of data covering a wide range of conditions measured as part of this work. From this comparison, a simpler and yet more accurate model has been developed in comparison with previously reported models in literature. This model demonstrates significant improvements over existing ones, particularly in predicting reaction behaviour under varying conditions. However, further work is required to account for the effects of methanol and water in the feed. A state-of-the-art global optimization algorithm, Covariance Matrix Adaptation, was employed, outperforming other tested algorithms. Additionally, careful data pre-processing minimized dataset bias and model parameter cross-correlation, contributing to a robust model. This study offers a more accurate model for designing reactors and could help in reducing risk in scaling up CO 2-to-methanol technologies, making them more practical for industrial use.
Download your free copy
Our publications are free to access. Simply provide your first name and email address to download.